How to Change Medium in Spheroid Cultures Without Losing Spheroids
Spheroid medium exchange is difficult for one simple reason: the biology is not attached. If aspiration is too fast, the tip is placed too centrally, or too much liquid is removed, spheroids can shift, break apart, or disappear into the pipette. To avoid this you should: aspirate gently, work at the well wall or exchange ledge instead of the center, and leave a defined residual volume rather than trying to remove every last microliter.[1] [3] [4]
For most labs, the safest way to change medium in spheroid cultures is to treat medium exchange as a controlled fluid-handling step rather than a simple pipetting task. That means using slow aspiration and dispense rates, keeping the plate stable, and choosing a residual volume that protects the spheroid even if it slightly reduces exchange efficiency. In specialized spheroid plates, lower residual volumes may be possible; in generic U-bottom or round-bottom ULA plates, a more conservative starting point is usually better.[1] [2] [4]
Why spheroids are often lost during medium exchange
Unlike adherent cell layers, spheroids are held in place mainly by gravity, well geometry, and their own structural integrity. That makes them sensitive to local flow, sudden suction, plate movement, and aggressive refill from above. Medium should be removed and added at the side of the well, not through a forceful central exchange step. In specialized spheroid plates such as Akura(TM) formats, dedicated ledge geometries are explicitly designed to make this easier and reduce the risk of aspiration-related loss.[3] [4]
A second common mistake is over-optimizing for maximum exchange instead of maximum retention. In practice, a slightly larger residual volume is often the right trade-off, especially for fragile, small, newly formed, or loosely compacted spheroids. Standard spheroid media-exchange guidance for 96-well round-bottom plates recommends starting conservatively, even around 50% residual volume, and optimizing from there; for more stable spheroids, a larger exchange fraction may become possible.[1]
A practical manual workflow for gentle spheroid medium exchange
Before starting, let the plate sit flat and undisturbed for a short moment so the spheroids are in their expected position. Then handle the plate smoothly and avoid abrupt movements. For non-adherent cultures, even transport can disturb the biology, so the less unnecessary motion, the better.
When removing spent medium, place the pipette tip at the well wall rather than in the center of the well. In plates with a dedicated exchange ledge, work from that ledge. In round-bottom or ULA plates without such a feature, stay high and lateral rather than deep and central. The goal is to remove liquid while keeping the flow path away from the spheroid itself.[3] [4]
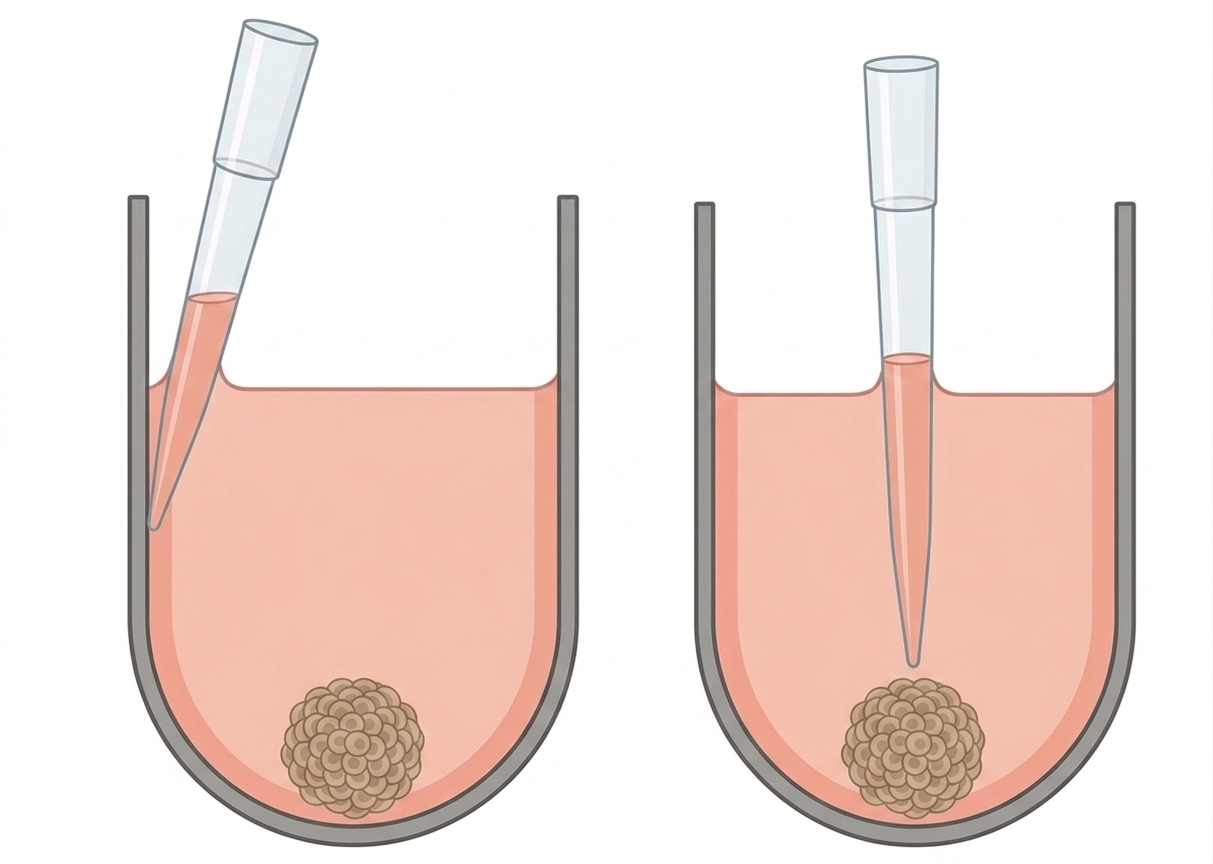
Aspirate slowly. For manual workflows, “slowly” matters more than “quickly but carefully.” Use low aspiration rates, and automation notes for spheroids recommend starting with the slowest practical aspiration and dispense settings, then adjusting only after confirming that the biology remains undisturbed.[2] [4]
Do not try to empty the well completely on the first attempt. Leave a residual volume that protects the spheroid from direct suction and reduces abrupt fluid motion near the culture compartment. In some spheroid plate systems, 10-20 µL is a reasonable conservative starting range for 96- and 384-well medium exchange, while specialized exchange geometries can support lower residual volumes. In standard round-bottom 96-well spheroid workflows, starting with a much larger residual fraction can be the safer choice until the exact behavior of the plate, medium, and spheroid type is known.[1] [2] [3]
When adding fresh medium, again work at the wall rather than dispensing directly onto the spheroid. Refill slowly enough that the incoming liquid climbs the wall and spreads into the well instead of hitting the culture as a jet. For Akura(TM) 384-well-plates, for example, the tip should be placed at the ledge for both removal and refill, with low aspiration and dispense rates to preserve the spheroid position.[2] [4]
Finally, keep the workflow consistent across wells. Medium exchange becomes unreliable when tip depth, angle, aspiration speed, refill speed, or residual volume vary from one column to the next. Even if the method “works,” inconsistent technique will usually show up later as well-to-well variability, unexpected assay noise, or unexplained outliers.
Starting points by plate type
| Plate type | Safest initial tip position | Residual volume strategy | Practical note |
|---|---|---|---|
| Standard 96-well round-bottom or ULA spheroid plate | Sidewall, away from center | Start conservatively; often much more residual is needed than in adherent-cell workflows | Optimize empirically for your plate and spheroid stability. |
| Standard 384-well spheroid workflow | Sidewall, very controlled depth | Leave more than you think you need at first; small wells are less forgiving | Use especially slow aspiration and refill. |
| Specialized spheroid plate with exchange ledge | Exchange ledge | Lower residual volumes may be achievable | In Akura(TM)-style workflows, low-speed ledge-based exchange is central to retaining spheroids. |
Common mistakes that cause spheroid loss
The most common error is placing the tip too close to the spheroid. In generic plates, that often means going too deep or too central. In specialized plates, it means ignoring the ledge and aspirating from the wrong position.
The second major error is aspirating too aggressively. Smaller or less compact spheroids usually require gentler settings than large, tight spheroids. What works for one model may be too harsh for another.
A third mistake is chasing near-complete exchange during a manual process. Higher exchange fractions can look attractive on paper, but if retention suffers, the workflow becomes biologically and analytically worse. A stable residual volume with high retention is usually better than an aggressive exchange step that causes variable tissue loss.
And finally do not treat all plates the same, well geometry matters. A method that behaves well in a plate with a dedicated exchange ledge may fail in a generic U-bottom or round-bottom format, and 384-well plates usually need tighter process control than 96-well plates.
When manual medium exchange reaches its limit
Manual medium exchange is often acceptable at small scale, especially during assay development or early feasibility work. But as plate counts rise, manual workflows usually become harder to standardize. Small differences in speed, tip depth, angle, and timing accumulate quickly, and those differences are amplified in fragile 3D cultures. Automated spheroid media exchange can help to improve reproducibility, reduce handling variability, and maintain gentle fluid movement over many repeated wells or plates.
That is the main reason automated systems are attractive in spheroid workflows: they do not just save time, they reduce operator-dependent variability. On the current WASH+ product page, we describe a WELLJET-based workflow that aspirates from the liquid surface, leaves a tunable residual volume, and dispenses along the well wall, with compatibility across 96- and 384-well plates and no physical WELLJET retrofit required. We show residual volumes down to 5 µL in Akura(TM) plates and typical processing times of about 2 minutes for 96-well plates and about 5 minutes for 384-well plates.
Conclusion
If you want to change medium in spheroid cultures without losing spheroids, the safest approach is usually not to maximize exchange, but to minimize disturbance. Keep the plate stable, aspirate at the sidewall or exchange ledge, use slow settings, leave a protective residual volume, and refill gently along the wall. Once that manual method is stable, you can optimize toward lower residual volume or higher throughput. If the workflow still depends too heavily on operator skill, that is usually the point where automation becomes worthwhile.
