How to Dispense Matrigel Domes in 96-Well Plates Without Premature Gelation
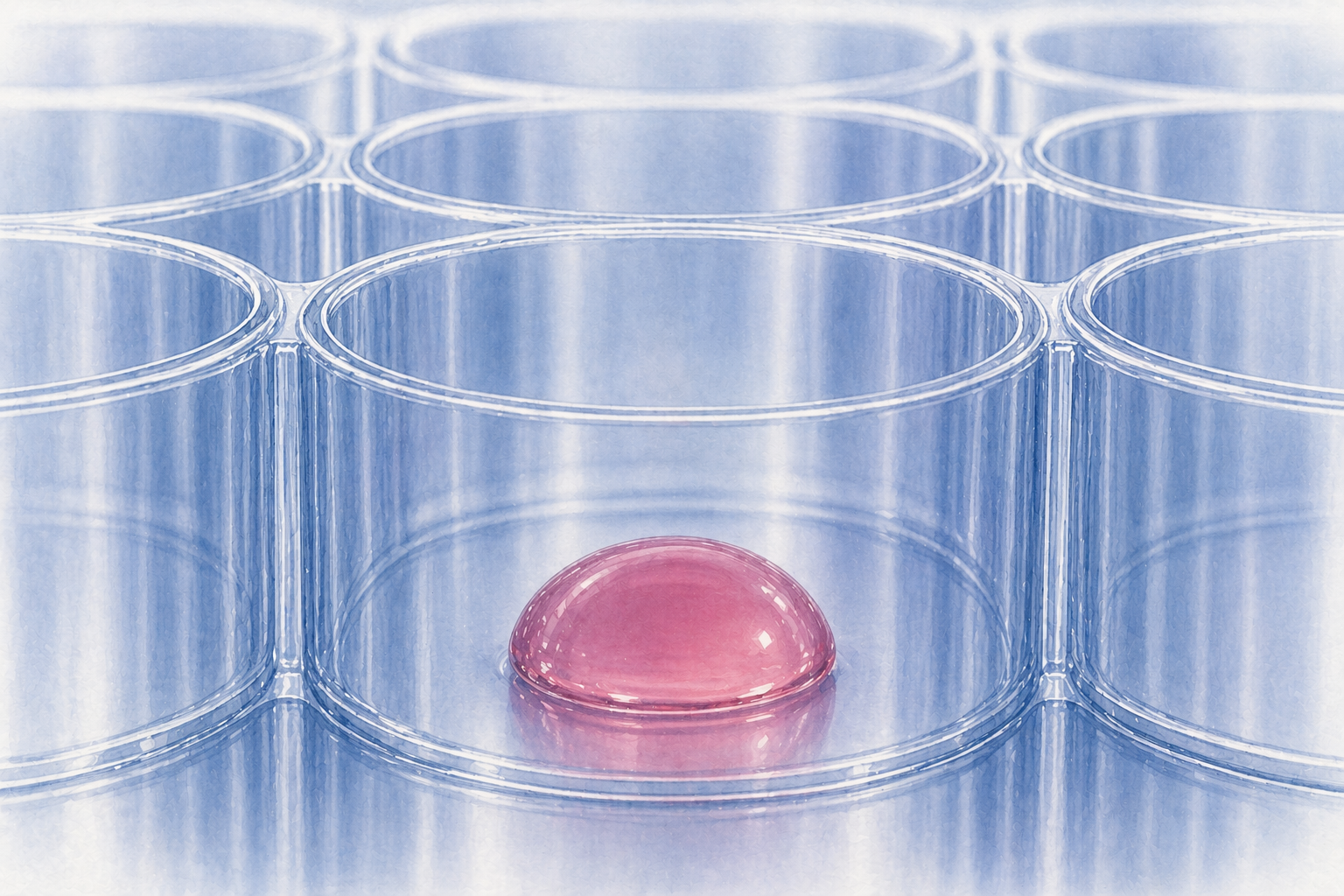
Matrigel dome dispensing is one of the most temperature-sensitive steps in many organoid and 3D cell culture workflows. The goal sounds simple: place a defined droplet of cold extracellular matrix on the bottom of each well, let it polymerize into a stable dome, and then add culture medium without disturbing the structure.
In practice, the workflow is often difficult to scale. Matrigel and related ECM hydrogels warm quickly in tubes, tips, reservoirs, and dispensing channels. As the material warms, viscosity changes, droplets become inconsistent, air bubbles appear more easily, and premature gelation can block tips or nozzles. This is especially problematic when moving from a few manual wells to full 96-well plates or multi-plate workflows.
This guide summarizes the key parameters for reproducible Matrigel dome formation and explains how cooled, low-dead-volume dispensing can make the workflow easier to automate.
What is a Matrigel dome?
A Matrigel dome is a small droplet of basement membrane matrix, often mixed with cells or organoid fragments, that is dispensed onto the bottom of a cell culture well and then allowed to polymerize. After gelation, culture medium is added around the dome. A good Matrigel dome is:
- centered on the bottom of the well
- compact and hemispherical rather than spread out
- free of visible air bubbles
- separated from the well wall
- consistent in volume across the plate
- stable enough to tolerate medium addition and later medium exchange
For many organoid workflows, dome volumes are in the low microliter range. The exact volume depends on the cell type, matrix concentration, well format, assay design, and imaging requirements.
Why Matrigel dome dispensing is difficult
Matrigel and similar ECM hydrogels are not standard aqueous reagents. They are viscous, biologically complex, and strongly temperature-sensitive. Small changes in handling can lead to large changes in dome quality.
- Premature gelationThe material warms before dispensing is complete. This can happen in the source tube, pipette tip, reservoir, tubing, pump head, or nozzle.
- Variable dome volumeViscosity changes during the run, making later wells behave differently from earlier wells.
- Flat or irregular domesThe droplet spreads across the bottom instead of forming a compact dome.
- Air bubblesBubbles can be introduced during mixing, aspiration, or dispensing. They can affect dome geometry and imaging quality.
- High reagent lossStandard dispensing systems may retain too much material in tubing, reservoirs, or cassettes. This is especially costly for expensive ECM reagents.
- Poor scalabilityA workflow that works manually for one plate may become unreliable when repeated across many plates.
The central challenge is to keep the material cold until it reaches the well, while still allowing controlled polymerization after the dome has been placed.
What a reproducible dome workflow needs
| Parameter | Why it matters |
|---|---|
| Temperature | Prevents premature gelation before dispensing |
| Dome volume | Defines assay geometry, diffusion distance, and imaging conditions |
| Tip or needle position | Determines whether the droplet stays centered or spreads |
| Dispensing speed | Affects droplet detachment, bubble formation, and dome shape |
| Dead-volume | Determines how much expensive ECM material is wasted |
For manual work, these parameters depend heavily on operator technique. For automated work, they must be built into the fluid path, cooling strategy, and dispensing geometry.
Manual Matrigel dome dispensing: practical workflow
The exact protocol should always be optimized for the cell type, matrix formulation, plate type, and downstream assay. The following workflow describes the general principles behind reliable dome dispensing.
1. Prepare the plate layout before thawing the matrix
Before bringing Matrigel into the workflow, decide:
- which wells will receive domes
- the dome volume per well
- the target number of cells or organoids per dome
- whether edge wells will be used
- how long the plate will remain outside the incubator
- how culture medium will be added after polymerization
This avoids unnecessary handling time once the matrix is thawed.
2. Keep the matrix cold until dispensing
Temperature control is the most important part of the workflow. The matrix should remain cold during thawing, mixing, aliquoting, and dispensing preparation.
Practical steps include:
- thawing according to the supplier protocol
- using pre-chilled tubes where appropriate
- keeping working aliquots on ice
- working with small batches instead of one large warm aliquot
- minimizing the time between loading and dispensing
- avoiding unnecessary pauses during the run
The important point is not only to cool the source tube. Every part that contacts the matrix can become a warm-up point.
3. Mix gently and avoid bubbles
Bubbles are difficult to remove once they are inside a viscous hydrogel. They can also affect the apparent dome volume and interfere with imaging.
To reduce bubbles:
- mix slowly and deliberately
- avoid vortexing cell-matrix mixtures
- use wide-bore tips if required for fragile organoids or large fragments
- avoid repeated aspiration and dispensing
- do not introduce air at the end of the pipetting stroke
- inspect the mixture before dispensing
If bubbles appear in the source material, it is often better to prepare a fresh aliquot than to continue with a compromised mixture.
4. Dispense to the center of the well bottom
For dome formation, the droplet should be placed at the bottom center of the well. If the droplet touches the wall, it may spread upward or form an asymmetric gel.
For manual pipetting:
- position the tip close to the well bottom
- avoid scraping the plastic
- dispense slowly and consistently
- keep the tip angle and depth consistent across wells
- withdraw the tip carefully to avoid pulling the dome upward
For small domes, even slight differences in tip position can cause visible variation across the plate.
5. Allow the dome to polymerize before adding medium
After dispensing, the dome must be allowed to solidify before culture medium is added. The polymerization conditions depend on the matrix, concentration, volume, plate type, and cell culture protocol.
Medium should be added gently along the wall of the well, not directly onto the dome. Direct flow onto the dome can detach or deform the gel, especially shortly after polymerization.
Common problems and how to fix them
Likely cause: The matrix is warming before or during dispensing.
What to change:
- reduce time outside cold storage
- work with smaller aliquots
- pre-cool contact surfaces where compatible with the protocol
- avoid pauses after loading
- reduce the number of wells dispensed per loading step
- consider cooled dispensing if running full plates or multiple plates
For automated dispensing, the source reservoir alone is not enough. The fluid path, pump area, and nozzle region can also warm the material.
Likely cause: The droplet spreads before polymerization or is dispensed with too much momentum.
What to change:
- reduce dispensing speed
- check that the tip or needle is centered
- use a more controlled dispense height
- confirm that the plate surface and matrix concentration match the protocol
- reduce vibration or plate movement after dispensing
- validate whether the plate should be cold or warm at the point of dome placement
Flat domes can also occur when the material has warmed and changed viscosity before reaching the well.
Likely cause: The droplet is not placed in the center, the volume is too large for the well geometry, or electrostatic forces pull it to the wall after placement on the bottom.
What to change:
- improve tip or needle alignment
- reduce dome volume
- use a plate format with more suitable well geometry
- avoid angled dispensing unless specifically validated
- ensure the plate is correctly seated in the holder
- de-ionize your plate before dome placement
This is especially important for imaging assays, where dome position affects focus, segmentation, and reproducibility.
Likely cause: Air is introduced during mixing, aspiration, or dispensing.
What to change:
- mix more gently
- avoid aspirating air at the end of the source volume
- use slower aspiration
- avoid repeated pipetting
- check whether the tip or nozzle retains droplets between wells
- remove visibly bubbled aliquots from the workflow
In automated systems, bubbles may also indicate poor priming, insufficient source volume, or air intake at the reservoir interface.
Likely cause: The matrix temperature, viscosity, or pressure conditions are changing over time.
What to change:
- shorten the run
- keep the working volume cold
- avoid long pauses
- reduce dead-volume in the fluid path
- verify channel-to-channel consistency
- use a dispensing setup designed for viscous, temperature-sensitive reagents
If the first wells and last wells look different, the workflow is usually not thermally stable enough.
Likely cause: The dispensing system has high dead-volume in tubing, reservoirs, or cassette channels.
What to change:
- reduce tubing length
- use smaller reservoirs
- avoid overfilling for small runs
- use a low-dead-volume fluidic path
- calculate required volume before starting the run
- include priming volume, residual volume, and safety margin in the calculation
For expensive ECM reagents, dead-volume can become one of the main cost drivers of the workflow.
When manual dispensing becomes the bottleneck
Manual dome dispensing can work well for small experiments. It becomes harder when the workflow requires full 96-well plates, multiple plates per run, small and consistent dome volumes, strict timing across wells, reduced operator-to-operator variation, integration into screening workflows, low reagent waste, documentation, and repeatability. At that point, automation becomes attractive. However, standard liquid handling automation does not automatically solve the problem. The system must be able to handle cold, viscous, temperature-sensitive material with low-dead-volume and controlled droplet placement.
Requirements for automated Matrigel dome dispensing
An automated Matrigel dome dispensing setup should provide:
1. Temperature control along the fluid path
The reservoir, tubing, pump region, and dispensing needles can all contribute to warm-up. Cooling only the source tube may not be enough for longer runs.
2. Low dead-volume
Hydrogels and ECM reagents are expensive. A dispensing workflow that requires milliliters of unused residual volume may be unsuitable for small batches or costly reagents.
3. Controlled bottom dispensing
For dome formation, the nozzle should place the droplet near the bottom center of the well. This is different from standard top dispensing into liquid-filled wells.
4. Consistent multi-channel behavior
If eight channels are used, each channel should deliver the same volume and droplet shape. Channel imbalance can create plate-position effects.
5. Simple setup and cleaning
A useful workflow should not require complex instrument modification, long setup times, or difficult cleaning steps between runs.
How COLD+ supports cooled Matrigel dome dispensing
COLD+ is designed for temperature-controlled dispensing of sensitive reagents on the Thermo Scientific Multidrop Combi. It combines three elements that are especially relevant for Matrigel dome workflows:
Passive cooling
COLD+ uses passive cooling elements for the reservoir and rotor area. The cooling modules are pre-cooled and then attached to the system without active refrigeration or modification of the Multidrop Combi.
The goal is to keep temperature-sensitive material below 10 °C during dispensing, reducing the risk of premature gelation inside the dispensing path.
Low dead-volume
COLD+ is based on the REAGENT+ low-dead-volume cassette concept. The fluidic path is designed to reduce residual volume compared with standard high-dead-volume setups. This is important for expensive ECM reagents where unused material can dominate the cost of a run.
Extended dispensing needles
COLD+ uses extended dispensing needles to place material closer to the well bottom. This supports defined dome formation in 96-well plates, instead of simply dispensing from above into the top of the well.
Together, these features make COLD+ suitable for workflows where Matrigel or related ECM hydrogels need to be dispensed cold, reproducibly, and with reduced reagent loss.
Example workflow concept for cooled Matrigel dome dispensing
- Pre-cool the COLD+ cooling modules.
- Prepare the Matrigel or cell-Matrigel mixture according to the lab protocol.
- Keep the working aliquot cold until loading.
- Attach the reservoir cooling module.
- Attach the rotor cooling module.
- Prime the low-dead-volume fluidic path.
- Dispense domes into the 96-well plate.
- Allow domes to polymerize under the required culture conditions.
- Add culture medium gently along the well wall.
- Clean the fluidic path according to the validated cleaning protocol.
This workflow should be validated for each matrix type, dome volume, cell type, and plate format.
COLD+ is not a replacement for protocol validation
Even with cooled dispensing, Matrigel dome workflows still require biological and technical validation across matrix lot, matrix concentration, cell or organoid density, dome volume, plate type, polymerization time, incubation conditions, medium addition method, imaging requirements, and downstream assay readout. COLD+ addresses the dispensing and temperature-control problem. It does not remove the need to optimize the biological protocol.
Practical checklist for Matrigel dome dispensing
Before starting the run:
- Plate map is finalized
- Dome volume is defined
- Matrix aliquot is thawed according to protocol
- Cell or organoid suspension is prepared
- Bubbles are avoided during mixing
- Working material is kept cold
- Tips, tubes, or fluidic path are prepared as required
- Dispensing position is validated
- Polymerization conditions are ready
- Medium addition method is defined
- Cleaning workflow is prepared
For automated workflows:
- Reservoir temperature is controlled
- Fluid path temperature is controlled
- Dead-volume is acceptable
- Nozzle position is validated
- Channel-to-channel consistency is confirmed
- First-well and last-well dome quality are comparable
- Run length does not exceed the validated cooling window
FAQ
Summary
Reproducible Matrigel dome dispensing requires more than accurate volume delivery. The material must remain cold before dispensing, bubbles must be avoided, the droplet must be placed precisely on the well bottom, and the workflow must be fast enough to prevent warm-up during the run.
For manual work, this requires careful timing and technique. For automated work, it requires a dispensing setup designed for temperature-sensitive hydrogels.
COLD+ combines passive cooling, low-dead-volume fluidics, and extended dispensing needles to support cooled Matrigel dome dispensing on a standard Thermo Scientific Multidrop Combi. This makes it possible to scale hydrogel dome workflows while reducing premature gelation and reagent waste.
